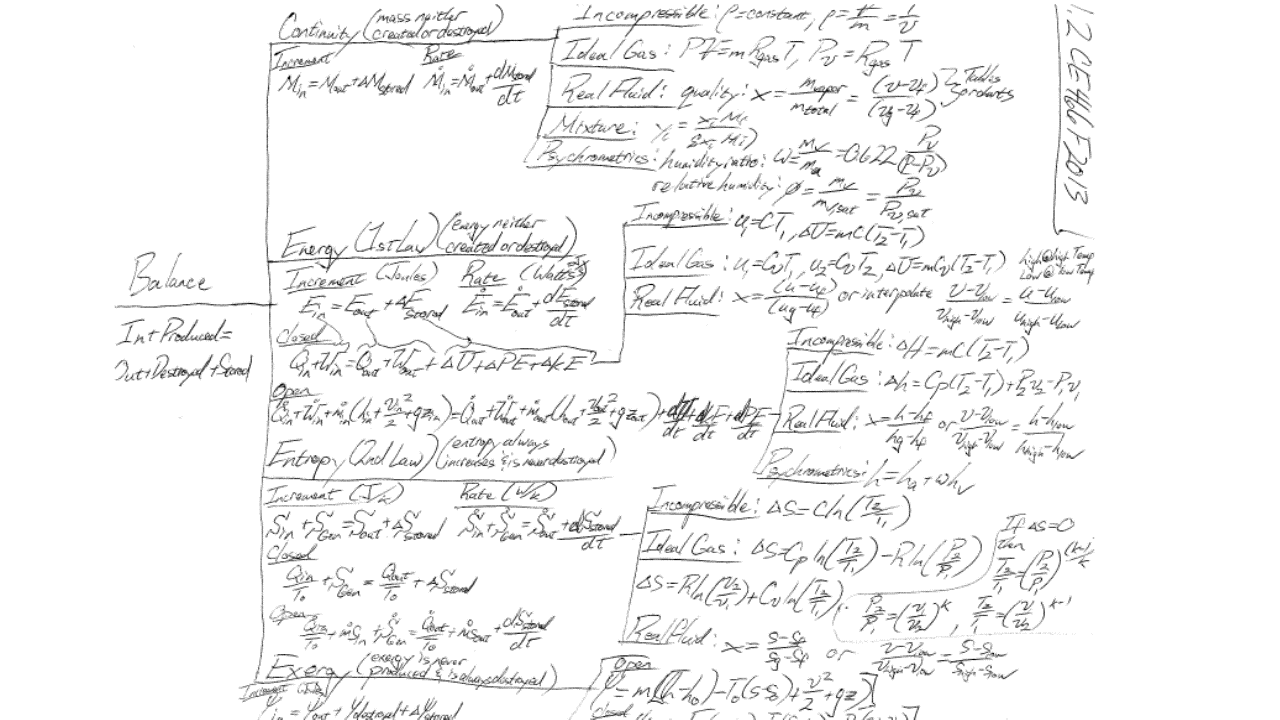ME 301 Thermodynamics
The Laws of the Universe. Once you know entropy, it’s irreversible.
Thermodynamics is a process. One of applying balances, fluid models, and software for solution and optimization. This process is shown by the following tree diagram, which encompasses all but the efficiency comparisons of ME 301.
Below I’ve posted all of my lecture notes, which borrow from my mentors Steve Penoncello (University of Idaho), John Pfotenhauer, Sanford Klein, and particularly Greg Nellis at the University of Wisconsin-Madison. These notes are intentionally old-school and hand written for a reason. Recent studies have shown that students retain information and perform better on exams when forced to write notes in their own personal narrative. The real magic though happens outside of class in real homework problems, often inspired by my experiences, and qualitative discussion prompts based on questions posed by students over the years. Students never forget some of the in-class demonstrations.
The combination of these methods has been successful. Student retention is now over 85%. Performance on the Fundamentals of Engineering (FE) Exam thermodynamics questions is 20% better than the national average. My course evaluations exceed 4.8/5.
Enjoy!
1st Law — Energy Balances
Energy is never created or destroyed so the first law of thermodynamics is simply a balance between energy forms. In this first part of class students learn to convert between energy forms — thermal, potential, kinetic, mechanical, heat, and work. Along the way we learn the basic property models for fluids — real, ideal, incompressible. We also learn the most basic form of efficiency as what you want divided by what you paid to get it.
Lesson 1: Introduction to ME 301
Lesson 2: Systems and Balances
Lesson 4: The Properties of Fluids
Lesson 5: Properties of Incompressible and Ideal fluids
Lesson 6: Closed System Balances
Lesson 7: Demonstrations of Closed System Balances
Lesson 8: Open System Balances
Lesson 9: Open, Un-steady systems
Lesson 10: Demonstrations of Open, Un-steady Systems
Lesson 11: Component Analysis Part 1
Lesson 12: Component Analysis Part 2
Lesson 13: In-class demonstrations and review
2nd Law — Entropy Balances
In the world of thermodynamics you never get back everything you put in because of entropy. In this second part of class students learn to balance entropy, which determines the direction and efficiency limits of processes.
Lesson 14: The 2nd Law and Direction
Lesson 15: Maximum aka Carnot Efficiencies
Lesson 16: Closed System Entropy Balance
Lesson 17: Entropy for Incompressible and Ideal Fluids
Lesson 18: Open System Entropy Balance
Lesson 19: Open System Entropy Balance Part 2
Lesson 20: Isentropic Efficiencies
Lesson 21: Isentropic Efficiencies Part 2
Lesson 22: Isentropic Efficiencies Part 3
Lesson 23: Maximum Work Available
Lesson 24: Exergy Analysis of Closed Systems
Lesson 25: Exergy Analysis of Open Systems
Lesson 26: Inventing Absolute Temperature Scales
Lesson 26: Entropy and Design
Thermodynamic Cycles
In this final third of class we apply the thermodynamic laws to size and design thermodynamic systems, specifically power and refrigeration cycles. We often find that the limitations are actually not due to the thermodynamic laws so much as practical design limitations. This presents many opportunities for innovation.
Lesson 28: Power Cycles — Concepts and Carnot
Lesson 29: Power Cycles — Rankine
Lesson 30: Power Cycles — Advanced Rankine
Lesson 31: Power Cycles — Brayton
Lesson 32: Power Cycles — Brayton Cycle for Propulsion
Lesson 33: Power Cycles — Internal Combustion Engines
Lesson 34: Power Cycles — Stirling
Lesson 35: Refrigeration Cycles — Concepts and Carnot
Lesson 36: Refrigeration Cycles — Vapor Compression
Lesson 37: Refrigeration Cycles — Heat Pumps
Lesson 38 Refrigeration Cycles — Refrigerant Selection
Lesson 40: Refrigeration Cycles — Advanced and Combined Cycles
Lessons 41-42: Course review and evaluation