I made these mistakes when I was learning. Just about every student in my lab has made them too. It’s all too common to have cryogenic copper confusion. It ends here.
The root of the confusion lies in the heat transfer promised land, as illustrated by the below chart of thermal conductivity of copper at cryogenic temperatures. An even better comparison than this chart is in Jack Ekin’s FANTASTIC book that is absolutely required reading for my lab: “Experimental Techniques for Low Temperature Measurement” Jack is so wonderful he’s even posted the figures openly available for people to access on-line and his thermal conductivity chart is here: http://www.researchmeasurements.com/figures/6-13.pdf
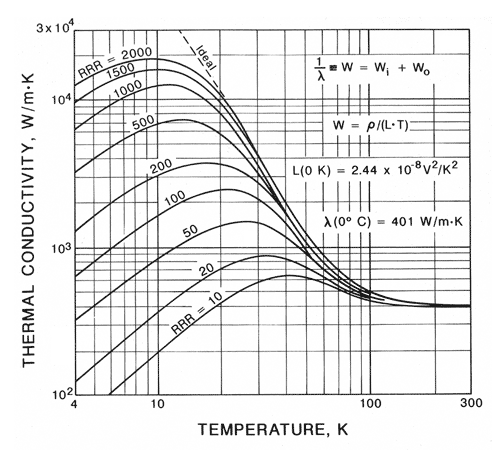
Yes you see that right. The thermal conductivity of copper varies two orders of magnitude at cryogenic temperatures. If you look at the figure in the link above from Jack Ekin’s book, RRR=2000 copper has a thermal conductivity at 10 K on par with the highest of diamond and sapphire. This thermal conductivity is a full 6!! orders of magnitude higher than some plastics. To put that into reference if you had a 10 K temperature gradient across a plastic bus bar, you’d have a 0.00001 K gradient with RRR=2000 copper. — The heat transfer promised land — and, like so many promised lands, has led many young cryogenicists to the school of hard knocks.
C110, C101, C102, ETP, OFHC, RRR???
All copper is not the same. If you go to common material suppliers your choices are typically C110, a.k.a. electrolytic tough pitch (ETP), or C101, a.k.a. oxygen free high-purity copper (OFHC). Looking back on the graph, both ETP and OFHC are listed next to a RRR of 50, and a full two orders of magnitude lower than RRR=2000 copper. Most copper scrap or tubing that’s sat around should probably be assumed RRR=25 or less. RRR stands for residual resistance ratio and is measured via the ratio of electrical resistance at 295 K versus immersed in liquid helium at 4 K. Electrical resistance depends primarily on purity and granularity of the sample. So if you by OFHC and carefully anneal it many times, you too can realize a RRR of 2000. But beware.
Once you remove all impurities and anneal copper to a RRR=2000, you’ve removed everything that made it strong. You’ve basically got a bar of soft material that behaves similar to lead or pure silver that you could scratch with your fingernail. This very high purity state is very attractive to impurities. People who seriously need high conductivity copper end up having to keep it sealed to prevent impurities via multiple platings or plastic wrap to prevent oxidation in air, because once it is in air, it’s no longer oxygen free high-purity. Moreover, this very soft material is hardly usuable for traditional metal applications because it has low strength. Further, it’s difficult to machine because it galls and grabs tooling. One of my students, who shall remain nameless for this post, decided to proceed with a C101 copper round because the machine shop had it and he was in a hurry. His piece ended up sitting for days immersed in an alum solution while he tried to dissolve away the tap he broke in it.
Calculating Thermal Diffusion
What’s potentially even more common than breaking tooling with copper is designing a part without doing simple heat transfer calculations to justify the design decisions. Problems are usually more time limited with cryogenics than temperature limited, which adds an additional dimension beyond traditional thermal conductivity. My co-advisor Greg Nellis and Sandy Klein’s classic text “Heat Transfer” is also required lab reading. Greg presents a handy equation for estimating the thermal diffusion time constant:
tau = L^2/(4*alpha)
where tau is the approximate time it takes for a thermal wave to propogate through a material of length L and thermal diffusivity alpha. Thermal diffusivity is the ratio of thermal conductivity to density times heat capacity. Jack Ekin’s book has a great graph on thermal diffusivity here: http://www.researchmeasurements.com/figures/6-3.pdf. With this you can quickly gauge whether, from a time perspective, it makes sense to have a higher purity material in your system. That 6 order of magnitude difference between copper and plastic changes a 10 minute equilibration time to a 19 year(!!) equilibration time.
Clever Geometry Hacks
One more trick to convince yourself that you don’t need high purity copper is geometry. A common problem in cryogenics is the routine calibration of temperature sensors over a large range of operation (3-120 K). Heat transfer occurs, by definition, through a temperature gradient. So one way to minimize temperature gradients, rather than going to a high conductivity material, is to reduce the heat transfer via geometry. By positioning temperature sensors in a “thermal dead-end” you minimize heat flow through the region and create a very uniform or equilibrium sector for sensor calibration. Again, Ekin’s wisdom shines through with this concept: http://www.researchmeasurements.com/figures/5-13.pdf.
The ultimate geometry hack is to remove the system from your cryostat entirely. An old saying in racing and aerospace, “any part left off the plane weighs nothing and never breaks.” You could extend this to include that it takes no design or machining time or expense either. The same goes for cryogenics.
In summary
You need to read Jack Ekin’s book and Greg Nellis’ text if you want to get good at cryogenics. The old saying goes, “A weeks worth of time in the library saves a year’s worth of time in the lab.” The same probably applies to simple design calculations and time spent on prototyping: do the simple calculation first to see what actually matters, then make your design decisions and keep them simple. Yes you could 3D print something complex these days and do an FEA analysis of heat transfer that looks pretty. But in the end, you’ll probably realize that the wonderful promised lands you had in mind may not be what you’re really looking for or needed.