Now that we have a framework for both social thermodynamics in equilibrium and in non-equilibrium transport we have an interesting opportunity to test the consistency of both through the time domain. This is enabled by the correlation between thermodynamic and transport properties — one of the greatest unsolved challenges in thermophysical properties is a direct derivation of transport properties from thermodynamic properties. Only recently has the residual entropy — the entropy that emerges due to real fluid intermolecular exchanges — been shown to be a powerful scaling tool to help with this challenge. This observation seams obvious in social space as the empathy that emerges during group exchange is powerful for efficient communication.
The diffusion properties directly compare thermodynamic and transport properties within a single variable. Through a juxtapose of thermal property trends with transport property trends, and comparing the combination, we may gain new insight on whether this framework transformation is remaining useful.
The Basics of Thermal Diffusion
One of the more interesting and important problems in my traditional cryogenics research is understanding how thermal diffusivity affects the time it takes a thermal wave to propagate through a material, also known as the thermal diffusion time constant. Thermal diffusivity (α) is a very interesting property that combines both thermodynamic properties (density ρ and heat capacity Cp) and a transport property (thermal conductivity k) via the equation:
α = k / ρ Cp
Thermal diffusivity, like all diffusion coefficients, has units of (m^2/s). From this you can calculate a parameter known as the thermal diffusion time constant (tau) for the time it takes a thermal wave to move through a length (L) through a bar of constant cross sectional area of uniform material via the equation:
tau= L^2/(4*α)
In other words, it’s the time it takes the change of some external condition (in this case temperature) to propagate through and be felt on the other side of something. With this equation you can quickly estimate the time it will take a system to respond to a step change in temperature at a boundary and it’s sensitive! For example, the difference in thermal diffusivity between copper and plastic at cryogenic temperatures changes a 10 minute equilibration time to a 19 year(!!) equilibration time — so many stories…
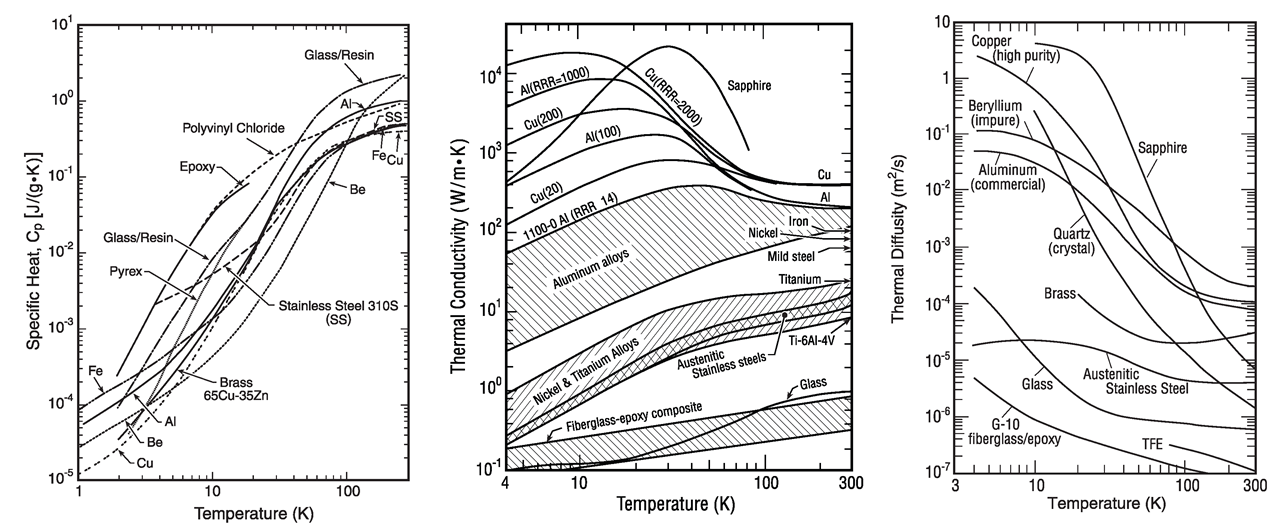
How can that happen? Here’s a figure that shows how heat capacity (left panel), thermal conductivity (center panel), control the thermal diffusivity (right panel). These are from Jack Ekin’s book, which I can’t recommend more highly, on cryogenic materials: http://www.researchmeasurements.com/figures/6-3.pdf. Plastics retain a large heat capacity at cryogenic temperatures because of the very long polymer chains with many small ways to store energy within the chain bonds. However, the thermal conductivity and ability to transfer that energy from one chain to another is very limited due to the irregularity of the chains. This combo leads to a low thermal diffusivity for plastics. Metals are much more simple and aligned with generally lower heat capacities, however the thermal conductivities can actually increase due to harmonic resonance (phonon) transport. This combo leads to a very high thermal diffusivity for pure metals.
What this means in the social domain
The book I’m compiling on social dynamics inspired by thermodynamics (here) has already established how values and temperature combine to cause the capacity to decrease with temperature (resources) and vMeme. My recent post on social transport mechanisms also shows that the number of diffusion mechanisms available decreases with temperature (resources) and vMeme. The question becomes the rate that these two parameters change relative to eachother with decreasing temperature. We don’t have equations of state that fix these trends yet. So we still need to use analogies.
A polymer chain molecule is analogous to a faculty member at a university — absolutely loaded with knowledge and information so as to yield a high capacity, perhaps so much so that it’s difficult for other molecules to relate and connect, in other words a low residual entropy/empathy and associated conductivity or transport value. This combination causes information and changes in boundary condition to diffuse incredibly slowly through the group.
A metal atom is analogous to a youth in a boarding or military school — relatively little knowledge (loaded with potential though!) so a fairly low capacity, however, is in a class among very homogeneous peers that know how to line up and speak on command, which are also united for a common cause (graduate). It’s like a crystal — very little room to move but when somebody says the head is mad, everybody knows what that means and fast. This leads to a higher residual entropy/empathy and associated conductivity or transport of info through phonon (acoustic vibration) resonance. This combination causes information and changes in boundary conditions to diffuse incredibly fast through the group, albeit over small ranges in temperature (resources).
Now let’s consider another class of material — high entropy metal alloys. This is a new class of materials emerging with very interesting properties for resilience. The closest material analogy in the above graphs would be stainless steel. You create these alloys by mixing many different atom types together, and creating crystal structures that maintain a balance between the constituent properties. The result? Although a decent heat capacity, a very low thermal conductivity that leads to a very low diffusivity close to plastics. The analogy that came to mind was the telephone game you play in elementary school where you start a message on one end of a line and watch how it changes when it comes out the other end. If you lined up a set of identical quadruplets and asked them to play the telephone game, you’d likely not have much of a game. Maximize the diversity of the group (analogous to a high entropy alloy) though and you’re bound for some fun. Which group, the quadruplets or the diverse group, are more likely to remain resilient in the face of an unknown stresser/challenge? My money’s on the diverse group — as long as the size of the group is not so large such that the slow information diffusion does not become the sensitive parameter.
(Note: this post is one chapter of what could become a book someday. The other chapters can be found here: https://hydrogen.wsu.edu/dr-jacob-leachman/ )